Engineers improve catalyst that destroys 'forever chemicals' with sunlight
Chemical engineers at Rice University have improved their design for a light-powered catalyst that rapidly breaks down PFOA, or perfluorooctanoic acid, one of the world's most problematic "forever chemical" pollutants. These pollutants aren't easily degraded and tend to linger in the environment. Mounting evidence suggests PFOA is harmful to human health.
Michael Wong and colleagues made the surprising discovery in 2020 that boron nitride, a commercially available powder that's commonly used in cosmetics, could destroy 99% of PFOA in water samples within just a few hours of exposure to ultraviolet light with a wavelength of 254 nanometers.
"That was great, because PFOA is an increasingly problematic pollutant that's really hard to destroy," said Wong, corresponding author of a study about the redesigned catalyst, published in Chemical Engineering Journal. "But it was also less than ideal because the boron nitride was activated by short-wave UV, and the atmosphere filters out almost all of the short-wave UV from sunlight. We wanted to push as much as possible boron nitride's ability to access energy from other wavelengths of sunlight."
PFOA is one of the most prevalent PFAS (perfluoroalkyl and polyfluoroalkyl substances), a family of compounds developed in the 20th century to make coatings for waterproof clothing, food packaging and other products. Wong said his team is assessing how well its composite photocatalyst works for breaking down other PFAS.
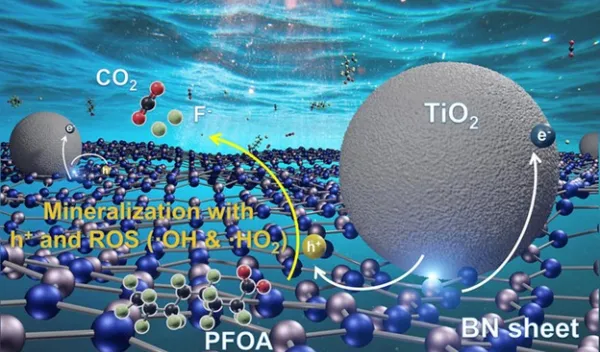
Long-wave UV, or UV-A, has wavelengths ranging from about 315-400 nanometers. It's what causes suntans and sunburns, and it's plentiful in sunlight that reaches Earth. Boron nitride is a semiconductor, and it isn't activated by UV-A. Titanium dioxide, a common ingredient in sunscreen, is a semiconductor that is activated by UV-A, and it has even been shown to catalyze the breakdown of PFOA, albeit very slowly, when exposed to UV-A.
Wong and study co-lead authors Bo Wang, Lijie Duan and Kimberly Heck decided to create a composite of boron nitride and titanium dioxide that married the best features of the individual catalysts. In their new study, they showed the composites powered by UV-A destroyed PFOA about 15 times faster than plain titanium dioxide photocatalysts.
By analyzing photocurrent response measurements and other data, Wong's team learned how its semiconductor composite harvested UV-A energy to break apart PFOA molecules in water. In outdoor experiments using plastic water bottles under natural sunlight, the team found the boron nitride-titanium dioxide composites could degrade about 99% of PFOA in deionized water in less than three hours. In salty water, that process took about nine hours.
Wong said the boron nitride and composite catalyst technologies have attracted attention from industrial partners in the Nanosystems Engineering Research Center for Nanotechnology-Enabled Water Treatment, which is funded by the U.S. National Science Foundation to develop off-grid water treatment systems.
"This team is advancing our understanding of how to remove harmful pollutants from water, even in low-resource or off-grid environments where access to clean drinking water remains a challenge," said Dana Denick, a program director in NSF's Directorate for Engineering.
